Innovating in the Brain Space
Neurolief’s Relivion® is a non-invasive, consumer medical device that treats headaches, migraines, and depression by stimulating branches of the occipital and trigeminal nerves. This stimulation modulates brain networks associated with pain and mood control.
Nekuda was called in on the project to bring full-stack expertise in usability, consumer-grade and medical-grade design, engineering for manufacturing and our know-how in head-mounted devices.
Benefits vs. Barriers
Strong and creative engineering was critical to this product — not just to elevate the technology, but more importantly to allow it to work correctly in the first place.
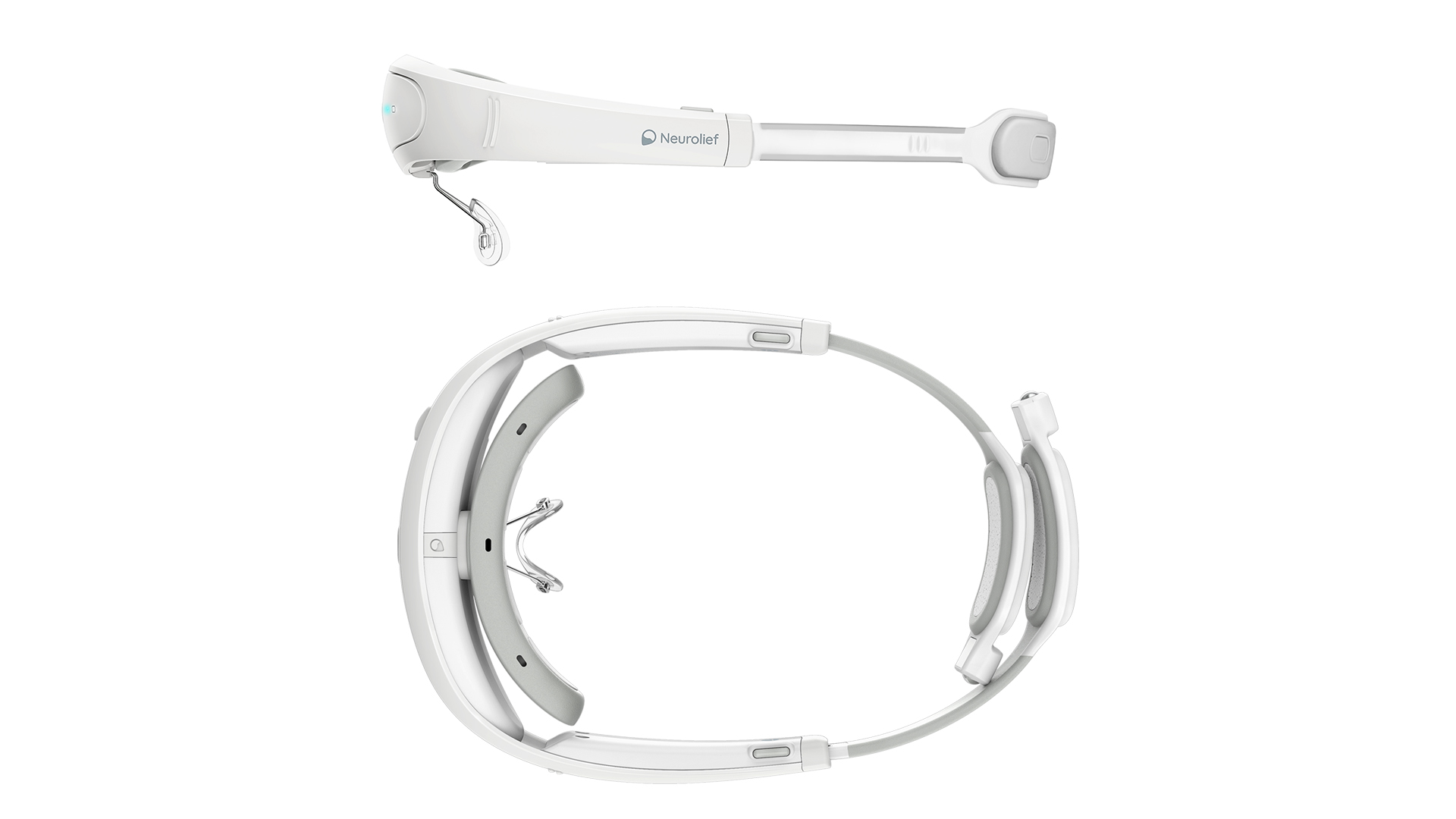
For the success of the device, it was essential to make sure that it fit a wide range of head sizes and shapes.
Another critical requirement was to provide reliable contact between the device and the user’s scalp, especially on the occipital area that is commonly covered with hair.
When it comes to devices like Neurolief’s, there are certain expectations from home users that shape a sensitive and accurate product definition.
Relivion’s edge case is the first-time user who is suffering from a migraine or depression. The challenge was to build a device that the user can put on and quickly adjust in order to get reliable skin contact on the first try.
The development process ran in parallel routes; one for supporting the clinical trials, and the other for the consumer-grade medical product.

Focused & Controlled Development
The entire project development process was focused, fast-paced, and extremely efficient, with clear cycles for validation and a continuous risk-mitigation program.
The convergence of consumer and medical design requires experience within medical regulatory frameworks along with an understanding of the consumer mindset.
Due to the self-administered nature of the Relivion and its treatment claims, the device had to be clinically proven as well as easy to wear. As a head-mounted product, it required special bio-compatible materials, sweat-wicking capabilities, frequent user-cleaning, and a high IP rating to prevent dust and water from entering the electronics. Our R&D process included a sequence of well-controlled usability models to test and qualify the ergonomics, fitting experience and proper usage supporting the use scenario and the relevant medical-grade requirements.
To achieve a clean, relaxed look-and-feel that would adhere to the FDA regulations for home devices, we had to hide the mechanical and electronic complexity of the device. Our team worked intensely to reduce the size, to organize the electronic components within the flowing ergonomic form, and to optimize structure and enclosure.
The Intersection of Consumer Medical
Today, medical technology teams are moving «beyond the pill» towards providing health rather than medications. Wearable devices like Neurolief’s Relivion is a big part of this trend.
We help our clients from these spaces to capture consumer-oriented insights and design holistically while delivering within a reasonable time to market.
Nekuda’s decade-long experience with consumer medical design allows us to provide an end-to-end process that helped Neurolief and many other clients create best-in-breed products and support them all the way from product definition through design and development, technology integration, to NPI and mass-production.

